Kenton D. Fong, M.D., Dean Hu, M.S., Shaundra Eichstadt, B.S., Deepak M. Gupta, M.D., Moshe Pinto, M.B.A., Geoffrey C. Gurtner, M.D., Michael T. Longaker, M.D., M.B.A., H. Peter Lorenz, M.D., Stanford and Sunnyvale, Calif.
Background:
Negative-pressure wound therapy is traditionally achieved by attaching an electrically powered pump to a sealed wound bed and applying subatmospheric pressure by means of gauze or foam. The Smart Negative Pressure (SNaP) System (Spiracur, Inc., Sunnyvale, Calif.) is a novel ultraportable negative-pressure wound therapy system that does not require an electrically powered pump.
Methods:
Negative pressure produced by the SNaP System, and a powered pump, the wound vacuum-assisted closure advanced-therapy system (Kinetic Concepts, Inc., San Antonio, Texas), were compared in vitro using bench-top pressure sensor testing and microstrain and stress testing with pressure-sensitive film and micro–computed tomographic scan analysis. In addition, to test in vivo efficacy, 10 rats underwent miniaturized SNaP (mSNaP) device placement on open wounds. Subject rats were randomized to a system activation group (approximately –125 mmHg) or a control group (atmospheric pressure). Wound measurements and histologic data were collected for analysis.
Results:
Bench measurement revealed nearly identical negative-pressure delivery and mechanical strain deformation patterns between both systems. Wounds treated with the mSNaP System healed faster, with decreased wound size by postoperative day 7 (51 percent versus 12 percent reduction; p < 0.05) and had more rapid complete reepithelialization (21 days versus 32 days; p < 0.05). The mSNaP device also induced robust granulation tissue formation.
Conclusions:
The SNaP System and an existing electrically powered negativepressure wound therapy system have similar biomechanical properties and functional wound-healing benefits. The potential clinical efficacy of the SNaP device for the treatment of wounds is supported. (Plast. Reconstr. Surg. 125: 1362, 2010.)
Disclosures:
K.D.F., D.H., and M.P. are current employees of Spiracur, Inc., but were not employed by Spiracur at the time of the animal experiments. M.T.L. and H.P.L. are scientific advisors to Spiracur, Inc., and have equity interests. The other authors have no financial interests to disclose.
From the Division of Plastic and Reconstructive Surgery, Department of Surgery, Stanford University School of Medicine, Stanford University; the Department of Bioengineering, Stanford University; and Spiracur, Inc. Received for publication October 13, 2009; accepted November 20, 2009.
Copyright ©2010 by the American Society of Plastic Surgeons
DOI: 10.1097/PRS.0b013e3181d62b25
Negative-pressure wound therapy for treatment of acute and chronic wounds has shown great efficacy, and numerous publications support its clinical use.1–6 In traditional negative-pressure wound therapy, a gauze or foam dressing directly contacts the wound bed, and an electrically powered pump is connected to a sealed enclosure over the wound. Mechanical stimulation has been theorized to be a key mechanism of action in negative-pressure wound therapy, leading to changes in biochemical signaling pathways, release of growth factors, and increased cellular proliferation.1,6–9 Saxena et al. described how negative pressure draws the wound surface into the open pores of the wound dressing foam, which creates regions of variable stress and strain.1 The wound surface in contact with the foamstruts is compressed, causing compressive stress and strain. Repeating patterns of high-strain gradients are created along the surface of the wound that appear as two-dimensional undulations across the wound surface.1 In addition, a fluidbased mechanism has been previously proposed as a contributing factor to the effectiveness of negative-pressure wound therapy. Negativepressure wound therapy removes excess interstitial fluid, leading to a decrease in interstitial pressure. Once the interstitial pressure drops below capillary pressure, capillaries are decompressed and are able to then reperfuse wound tissue.6 Thus, an effective negative-pressure wound therapy device must deliver mechanical stimulation and handle wound fluid dynamics within an appropriate negative-pressure range.
Recently, a novel ultraportable negative-pressure wound therapy system called the SNaP System was developed. The SNaP System consists of five basic elements: the vacuum/exudate cartridge, activation/ reset key, hydrocolloid dressing layer, extension tubing, and a gauze wound interface layer. Figure 1 shows photographs of the SNaP System (generation 1.0) used in this study. The commercially available version (generation 2.0) of the SNaP System has been further refined and simplified and can be seen in Figure, Supplemental Digital Content 1, https://links.lww.com/PRS/A155 (the yellow –75 mmHg model is shown). Compared with the earlier version of the device, there was an elimination of the button system, streamlining of the cartridge design, the addition of a red cartridge full/air leak indicator, and development of a customized hydrocolloid dressing layer. To create negative pressure without an electrically powered pump, a set of specialized constantforce springs causes forced air expansion within the system. A predetermined level of negative pressure is delivered in a constant fashion as exudate collects. With exudate inflow, the unique spring system moves the piston and expands the system volume to maintain the desired amount of negative pressure delivered to the wound. The disposable cartridge is produced with three different preset pressure levels (approximately –75, –100, and –125 mmHg), and is small enough to be worn on a patient’s leg, arm, or belt and hidden under clothing. Once placed on the patient, the SNaP System is left in place between dressing change visits and continues to deliver negative pressure unless the cartridge fills with exudates or there is an air leak. If the patient has a highly exudative wound that fills the capacity of the cartridge, the patient can change the cartridge at home. This type of negative- pressure wound therapy delivery system has several potential advantages over traditional pumps, including increased mobility, silent operation, and decreased cost.
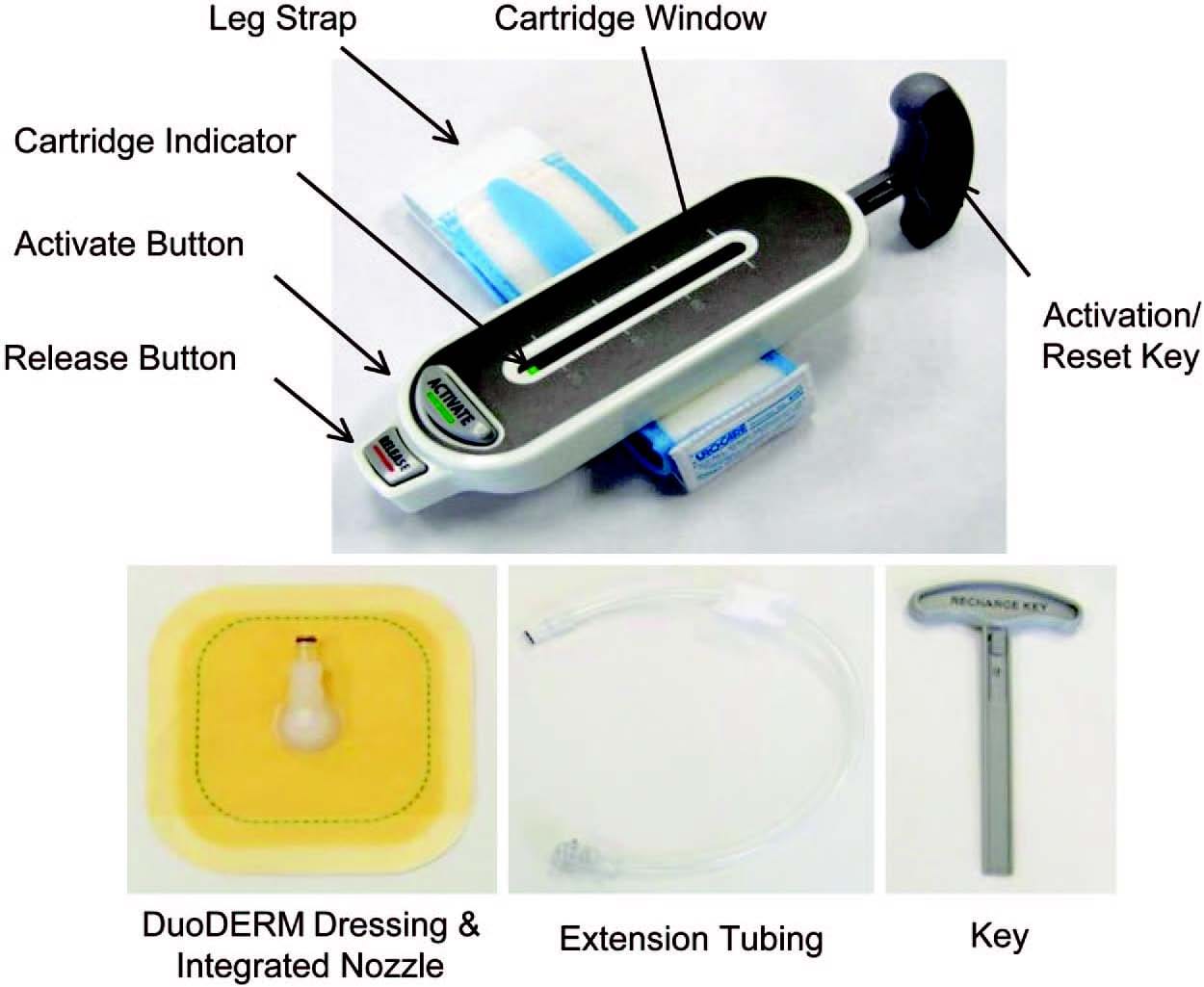
Fig. 1. The ultraportable SNaP Wound Care System (generation 1.0). When the activation button is depressed, negative pressure is delivered from the retraction of the sliding seal by constant-force springs to deliver a predetermined level of negative pressure.
This study compared the SNaP System to a benchmark negat
ive-pressure wound therapy product, the wound vacuum-assisted closure advanced- therapy system device (Kinetic Concepts, Inc., San Antonio, Texas), an electrical diaphragm- pump system. The vacuum-assisted closure device creates negative pressure by removing mass from the system and uses foam instead of gauze as the wound interface layer. The vacuumassisted closure device uses continuous pumping to compensate for pressure loss from pump backstreaming and wound exudate inflow. This study examined the negative pressure delivered by both systems in a static state and with wound exudate present. The mechanical stress and strain patterns produced at the wound bed by each system was also compared. To evaluate the effect of negative-pressure wound therapy delivered by the SNaP System on wound healing in an in vivo model, a miniaturized version of the SNaP System (mSNaP System) was created and tested in a rat open wound model developed by Isago et al.3 Using a vacuum-assisted closure system in their model, Isogo et al. found significantly smaller wound areas in animals treated with –50, –75, and –125 mmHg of pressure compared with those treated with no pressure or –25 mmHg of pressure.3
This study used a foam product for both the vacuum-assisted closure and SNaP System experiments solely for purposes of comparison of operation of the two systems. Because both electrical diaphragm-pump and forced expansion mechanisms generate negative pressure at the wound bed, we hypothesized that the end effects of negative pressure (i.e., exudate evacuation, mechanical deformation, mechanical stimulation of the wound, and ultimately faster wound healing) should be the same.
Materials and methods
Biomechanical Testing
Pressure Measurement
A simulated wound-dressing enclosure was constructed by placing a 6 x 6 x 3-cm piece of GranuFoam (Kinetic Concepts) on a polycarbonate plate. The foam was covered with a dressing layer fitted with a pressure delivery port, connected to either a vacuum-assisted closure device or SNaP System set to deliver –125 mmHg of negative pressure [see Figure, Supplemental Digital Content 2, which demonstrates the biomechanical pressure testing setup of the SNaP System without (above) and with (below) exudate simulation, https://links.lww.com/PRS/A156 (a, negative pressure provided by either vacuum-assisted closure (above) or the SNaP System (below) at 125 mmHg; b, pressure delivery port; c, dressing; d, foam contact layer; e, data-logging manometer; f, simulated exudates infused at 5 cc/hour)]. The underside of the plate was equipped with a separate port where fluid could be introduced into the system; a datalogging manometer was connected to monitor pressure at the underside of the foam. The pressure delivery system was activated and pressure level was logged every 20 seconds (4000 over 24 hours = 4000/24/60/60 = 0.0463 per second = 1 every 20 seconds) for 24 hours.
The above procedure was repeated with the addition of simulated wound exudate, a mixture of 50 percent glycerol and 50 percent water (see Figure, Supplemental Digital Content2B, which shows a schematic of this set-up, https://links.lww.com/PRS/A156). The mixture was loaded into a 60-cc syringe pump (New Era Systems, Wantagh, N.Y.) connected to the underside port of the test plate and infused at 5 cc/hour. The vacuum-assisted closure device or SNaP System was connected and activated at –125 mmHg, and pressure measurements were recorded every 20 seconds for 8 hours. The pressure delivery systems were massed (in the case of the vacuum-assisted closure device, only the collection canister was massed) both before and after the testing period to measure the amount of exudate collected.
Mechanical Stimulation
Both contact stress and strain were evaluated for the vacuum-assisted closure device and SNaP System. To evaluate contact stress, a simulated wound bed was created by laying a sheet of pliable thermoplastic elastomer onto a polycarbonate test surface. A piece of PressureX Micro pressure-sensitive film of 2 x 2 cm (Sensor Products, Inc., Madison, N.J.) was placed on top of the thermoplastic elastomer. GranuFoam (6 x 6 x 3 cm) was placed on top and the wound was sealed and connected to either a vacuum-assisted closure device or SNaP System [see Figure, Supplemental Digital Content 3, which shows the experimental setups for evaluating wound contact stress and for micro– computed tomographic wound surface strain imaging, https://links.lww.com/PRS/A157 (a, negative pressure provided by either vacuum-assisted closure (above) or SNaP System device (below) at 125 mmHg; b, pressure delivery port; c, dressing; d, foam contact layer; e, pressure-sensitive film; f, simulated tissue; g, simulated wound enclosure (above, left) inside micro–computed tomographic scanner)]. Negative pressure (either –125 mmHg or –75 mmHg) was applied for 120 seconds and the pressure-sensitive film was analyzed. Four different setups were performed: vacuum-assisted closure device at –75 mmHg, vacuum-assisted closure device at –125 mmHg, SNaP System at –75 mmHg, and SNaP System at –125 mmHg.
Contact strain produced by the vacuum-assisted closure device and SNaP System was evaluated using micro–computed microtomographic scanning, following a protocol similar to that developed by Saxena et al.1 A simulated wound-dressing enclosure was constructed inside a polyurethane block 5.5 cm in diameter, and a 3x3x1-cm sheet of thermoplastic elastomer was placed inside the block. On top of this sheet, a piece of 3 x 3 x 1-cm GranuFoam was placed. A dressing with connection tubing was then applied. The enclosure was then placed on the imaging bed of an Imtek MicroCAT II CT/MicroSPECT Scanner (Siemens Corp., New York, N.Y.) with connection tubing leading outside the scanner (see Figure, Supplemental Digital Content 3B, which shows a schematic of this setup, https://links.lww.com/PRS/A157). The tubing was connected to either a vacuum-assisted closure device or SNaP System set to produce either –125 mmHg or –75 mmHg of negative pressure, or to no pressure source. The imaging bed was positioned and a scan of the enclosure was acquired at a resolution of 36 x 36 x 41 μm. Five scans were obtained: no negative pressure, vacuum-assisted closure at –75 mmHg, vacuum-assisted closure at –125 mmHg, SNaP System at –75 mmHg, and SNaP System at –125 mmHg. Scans were reconstructed in real time using MicroCAT software and imported into AMIRA (Visage Imaging, Carlsbad, Calif.) for processing. From the threedimensional data set, slices parallel and perpendicular to the simulated wound bed were extracted. Simulated tissue surfaces were examined for evidence of deformation (strain) patterns under the different conditions.
Animal Studies
All animal experiments were performed under the Stanford University Animal Procedures and Care Committee’s approved protocol and were compliant with the guidelines specified in the National Institutes of Health’s Guide for the Care and Use of Laboratory Animals. All animals were housed in the Veterinary Service Center at Stanford Medical Center with 12-hour light/dark cycles and ad libitum water and rodent chow throughout the study period.
Study Groups
Ten Sprague-Dawley rats weighing between 200 and 250 g (similar in size and age to those used by Isago et al.3) were used in this study, divided randomly into two groups. The first group had the mSNaP System placed on their wounds with activation of negative pressure at approximately –125 mmHg. The second group had the system placed on their wounds without activation of negative pressure.
mSNaP System Negative-Pressure Wound Therapy and Sealant System
The mSNaP System used for animal testing is shown in Figure, Supplemental Digital Content 4, https://links.lww.com/PRS/A158 (A, constant-force spring; B, modified syringe body; C, activation valve; D, syringe plung
er seal; E, hydrocolloid skin dressing; F, on activation, the constant force springs pull on the plunger, maintaining a constant level of reduced pressure in the system as exudates collect). The system consists of a modified 7-cc Epilor syringe (part no. C3601; Qosina, Edgewood, N.Y.) with an activation valve at one end and a constant-force spring connected to a plunger. The syringe chamber is charged by evacuating air using a 20-ml syringe and closing the activation valve to lock the plunger in the charged position. GranuFoam was used as the contact layer to the wounds. A specially modified hydrocolloid dressing (DuoDERM; ConvaTec, Skillman, N.J.) was used as a dressing. The hydrocolloid dressing had a nozzle integrated into it using a washer system and ultraviolet light–cured epoxy (Loctite 3311; Henkel, Düsseldorf, Germany). After activation, a valve was attached to the nozzle dressing. The valve was then opened to create air/fluid communication, thus delivering negative-pressure wound therapy to the wound. Bench testing of the mSNaP System demonstrated it capable of delivering –125 ± 10 mmHg of negative pressure.
Surgical Procedure
A surgical wound-healing model was performed as described previously by Isago et al.3 Briefly, the animals were anesthetized and their backs were shaved and depilated. Wounds (2.5 x 3.0 cm) were excised through skin and panniculus carnosus using a scalpel and surgical scissors. The wounds of each animal were photographed and measured along the vertical and horizontal lengths of the body axis. Wounds were dressed with an interface layer of GranuFoam with a hydrocolloid cover sheeting, followed by connection of the mSNaP System. Mastisol ointment (Ferndale Laboratories, Ferndale, Mich.) was applied to intact skin of the animals to improve adhesion of the dressing. The hydrocolloid dressing edges were reinforced further using large Tegaderm dressings (3M, St. Paul, Minn.). The syringe portion of the system was fixed to the animals’ bodies by half-inch paper tape. Dressings were checked every 24 hours for all animals, with emptying and recharging of the system for those randomized to the system activation group. Wound closure was defined as complete reepithelialization without drainage and was evaluated on a daily basis after discontinuation of SNaP treatment by a plastic surgeon.
Histologic Evaluation
All dressings were removed on postoperative days 4 and 7. Punch biopsy specimens (6 mm) of the central portion of the wound bed were harvested and fixed in 4% paraformaldehyde. Animals were not killed to obtain punch biopsy specimens of wounds. Routine hematoxylin and eosin staining was used to examine granulation tissue formation.
Statistical Analysis
To assess group differences, a two-sample t test (two-tailed) or modified t test for uneven variance was applied. The values were considered to be significant at a level of p < 0.05.
RESULTS
Mechanical Testing Data
Similar Pressure Delivered by the SNaP System and the Vacuum-Assisted Closure Device When No Exudate Is Present
When set at –125 mmHg and tested over a period of 24 hours, the vacuum-assisted closure device delivered an average pressure of –124.9 ± 1.3 mmHg. The SNaP System delivered –120.7 ± 1.18 mmHg. Both systemsmaintained a steady level of negative pressure throughout the test period, with very small degrees of variability, as demonstrated in Figure 2. The pressures were measured over the duration of the test (4000 time point measurements). The stated standard deviations were calculated from all data points taken during the respective tests and reflecthowwell the device (vacuum-assisted closure advanced-therapy system or SNaP Cartridge) maintained a steady-state pressure.
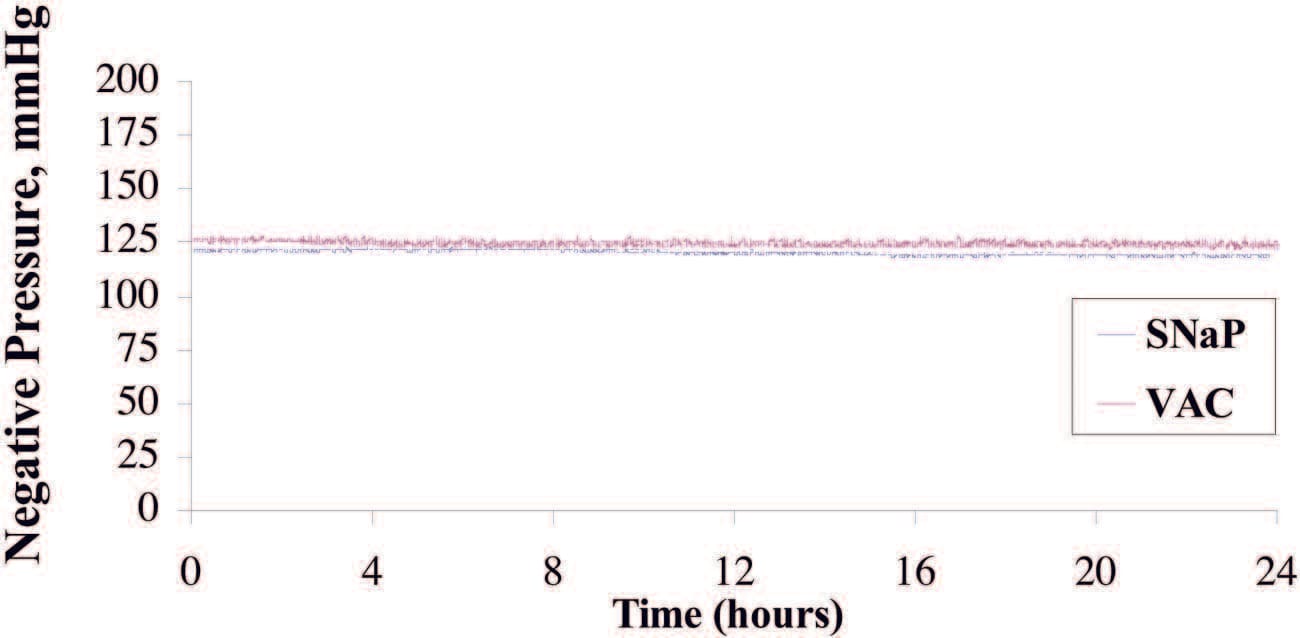
Fig. 2. Plot of pressure delivered under static state for SNaP System and vacuumassisted closure (VAC). The vacuum-assisted closure device delivered an average pressure of –124.9±1.3mmHg,whereas theSNaPSystem delivered –120.7±1.18mmHg. Both devices were able to maintain a steady level of negative pressure throughout the test period and demonstrated a very small degree of variability.
Similar Pressure Delivered by the SNaP System and the Vacuum-Assisted Closure Device When Exudate Is Present
Eight hours of pressure measurements were recorded during the infusion of simulated exudate at the rate of 5 cc/hour. The average pressure delivered by the vacuum-assisted closure device was 123.2 ± 0.8 mmHg, compared with 121.7 ± 3.1 mmHg delivered by the SNaP System. The pressure signal of the SNaP System, although confined to a tight band of negative pressure, exhibited a sawtooth pattern characteristic of minute movements of the constant-force spring mechanism to correct for pressure loss during exudate entry. A total of 40 cc of exudate was infused into the simulated wound during the duration of the test. The vacuum-assisted closure device collected 31 g, whereas the SNaP System collected 35 g of exudate. Figure 3 demonstrates that both systems delivered and maintained a steady level of negative pressure under conditions of fluid introduction. The pressures were measured over the duration of the test. The stated standard deviations were calculated from all data points taken during the respective tests and reflect how well the device (vacuum-assisted closure advanced-therapy system or SNaP Cartridge) maintained a steady-state pressure.
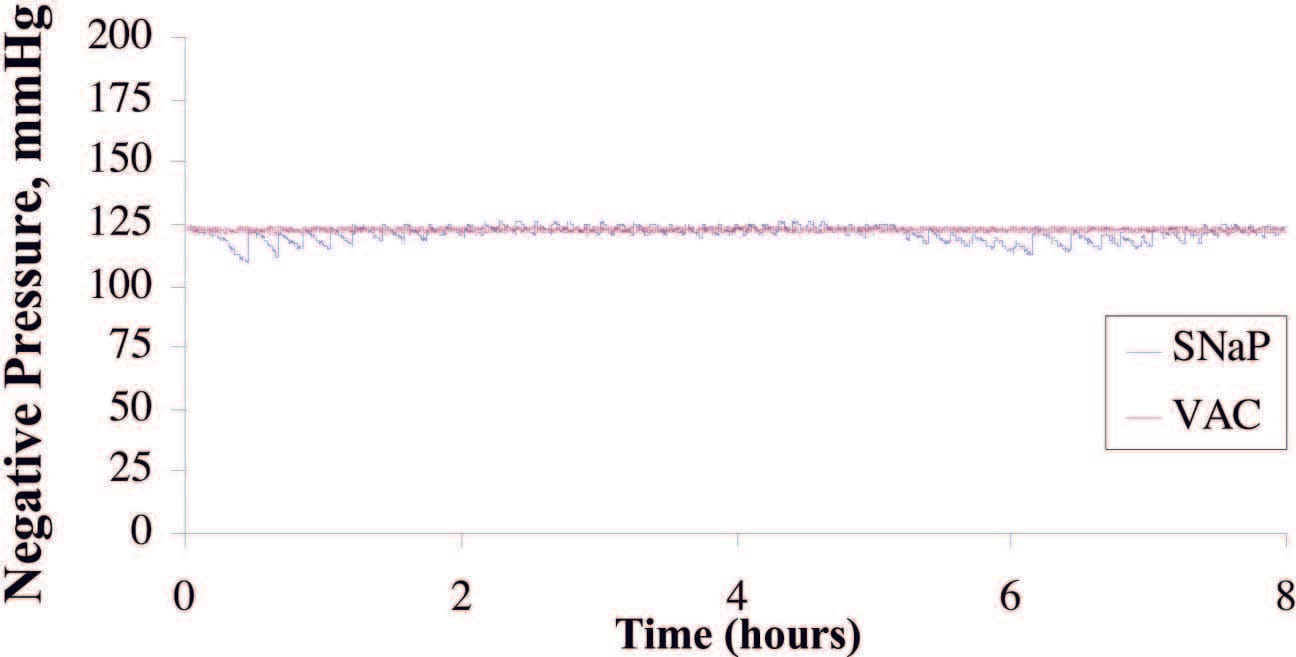
Fig. 3. Plot of pressure delivered during exudate test for SNaP System and the vacuum- assisted closure (VAC) device. Average pressure delivered by the vacuum-assisted closure device was –123.2 ± 0.8 mmHg; the SNaP System delivered –121.7 ± 3.1 mmHg. Both devices were able to deliver and maintain a steady level of negative pressure, even under conditions of fluid introduction.
Similar Contact Stress Patterns Delivered by the SNaP System and the Vacuum-Assisted Closure Device
The SNaP System and vacuum-assisted closure device produced similar contact stress profiles for equivalent pressure levels, as measured by micropressure- sensitive film (Fig. 4). The patterns of contact stress demonstrated that both systems conducted negative pressure through the foam and produced very similar wound surface stress gradients. The pressure distribution image is qualitative, showing relative areas of high stress and low stress. Further quantitative analysis of the data was precluded by the sensitivity limit of the pressuresensitive film used.

Fig. 4. Biomechanical testing, pressure measurement. (Left and second from left) Raw images of pressure-sensitive film recovered from under foam for vacuum-assisted closure (VAC) and the SNaP System at –75mmHgand –125 mmHg. Dark regions denote areas of high compressive stress in contact with film, and light regions denote no contact. Note that both the SNaP System and the vacuum-assisted closure device produce characteristic repeating patterns on length scales, consistent with foam pore size. (Right and second from right) Color-enhanced detail from pressure-sensitive films.
Similar Contact Strain Patterns Delivered by the SNaP System and the Vacuum-Assisted Closure Device
Notable features of the micro–computed tomographic scans are shown in Figure 5. This figure shows the foam microstructure to be clearly visible. Without delivery of negative pressure, the simulated wound bed/foam interface is smooth. Wit
h delivery of negative pressure at –75mmHgor –125 mmHg by either the SNaP System or the vacuumassisted closure device, the characteristic pattern of deformation that occurs with negative pressure occurs at the simulated wound bed/foam interface. This pattern matches the predicted stress and strain pattern of undulation described and demonstrated by Saxena et al.1 No discernible difference in the microdeformation patterns was observed between the two pressures tested for either the SNaP System or vacuum-assisted closure– treated experiments. Furthermore, the patterns of deformation appeared nearly identical for both the SNaP System and vacuum-assisted closure at the pressure levels tested. Because this was a qualitative measure, no further quantitative analysis was performed.

Fig. 5. Biomechanical testing, mechanical stimulation. Micro–computed tomographic scans parallel to the simulated tissue surface (top images, horizontal plane in reference image) and perpendicular (bottom images, vertical plane in reference image) for all tested pressure configurations. Note that both theSNaPSystemand the vacuum-assisted closure device produce similar characteristic repeating patterns at both pressures.
Animal Testing Data
Wounds Healed Faster with the Activated mSNaP System
Animals treated with themSNaPSystem had a 51 percent reduction in wound size compared with a 12 percent reduction in wound size of control subjects at 7 days (p < 0.05) (Fig. 6). Complete reepithelialization also occurred faster than in control subjects (21 days versus 32 days; p<0.05), as shown in Figure 7. See Figure, Supplemental Digital Content 5, which shows representative wounds at postoperative days 0 and 7, https://links.lww.com/PRS/A159 (note the increased granulation tissue and smaller size of the activated system–treated wounds compared with atmospheric controls). Comparison of animals treated with the mSNaP System to data from the study by Isago et al. using the vacuum-assisted closure device in the same animal model reveal very similar results. ThemSNaPSystem–treated wounds demonstrated a 51 percent decrease in wound size, which is comparable to the reported 40 percent decrease in wound size observed for vacuum-assisted closure–treated animals at 1 week.3 Control animals treated with atmospheric pressure in this study and the study by Isago et al. were 12 percent and 14 percent surface area size reduction, respectively.3
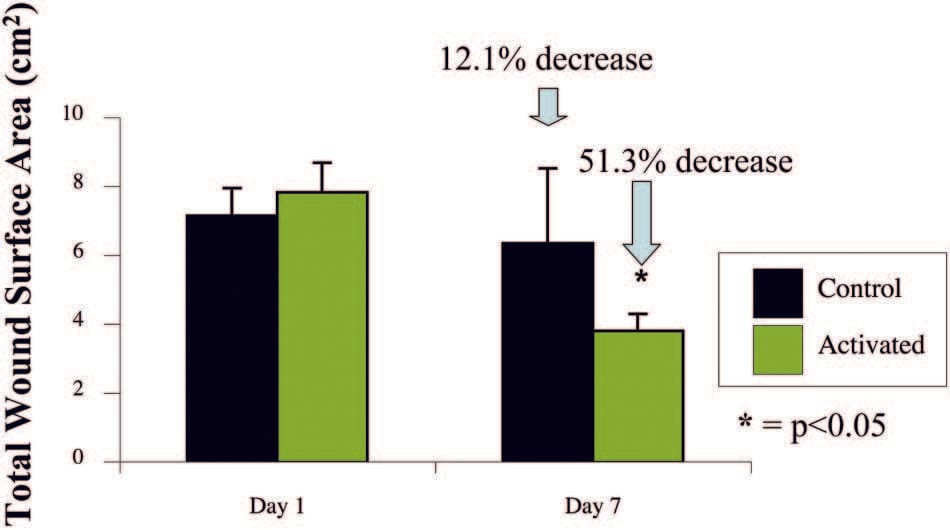
Fig. 6. The SNaP System decreases wound size. Negative-pressure wound therapy–treated animal wounds were significantly smaller than control wounds. Treated animals had 51 percent smaller wounds compared with 12 percent smaller wounds in control subjects on postoperative day 7 (p < 0.05).
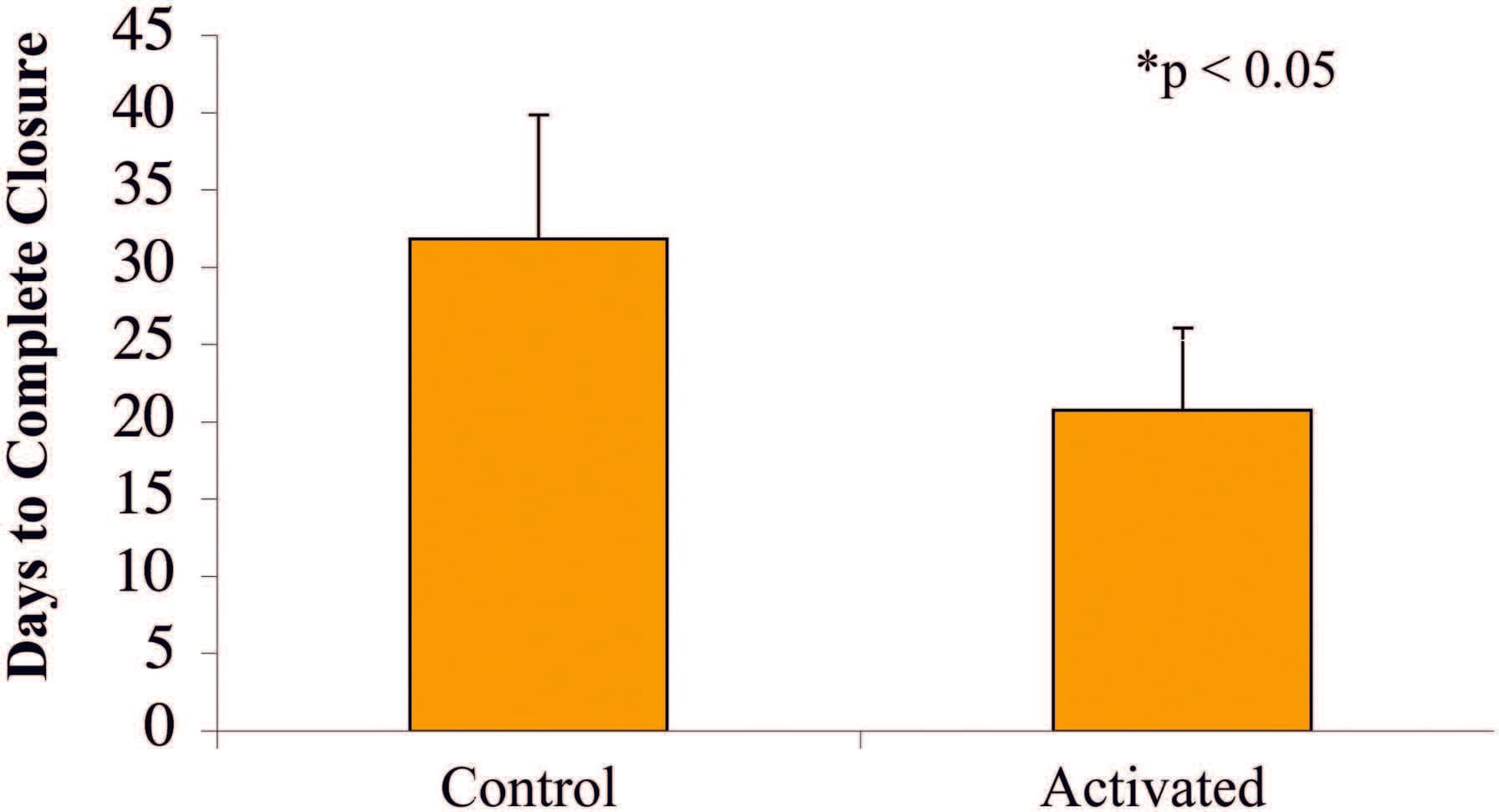
Fig. 7. Wounds treated with the SNaP System for 7 days healed faster (mean, 21 days) than control animal wounds (mean, 32 days) (*p < 0.05).
Wounds Had Greater Granulation Tissue with the Activated mSNaP System
Similar to previous reports for the vacuumassisted closure device,10 the mSNaP System promoted granulation tissue formation compared with controls, noted by gross examination of the wounds (see Figure, Supplemental Digital Content 5, https://links.lww.com/PRS/A159) and by hematoxylin and eosin staining on postoperative days 4 and 7 (Figs. 8 and 9).
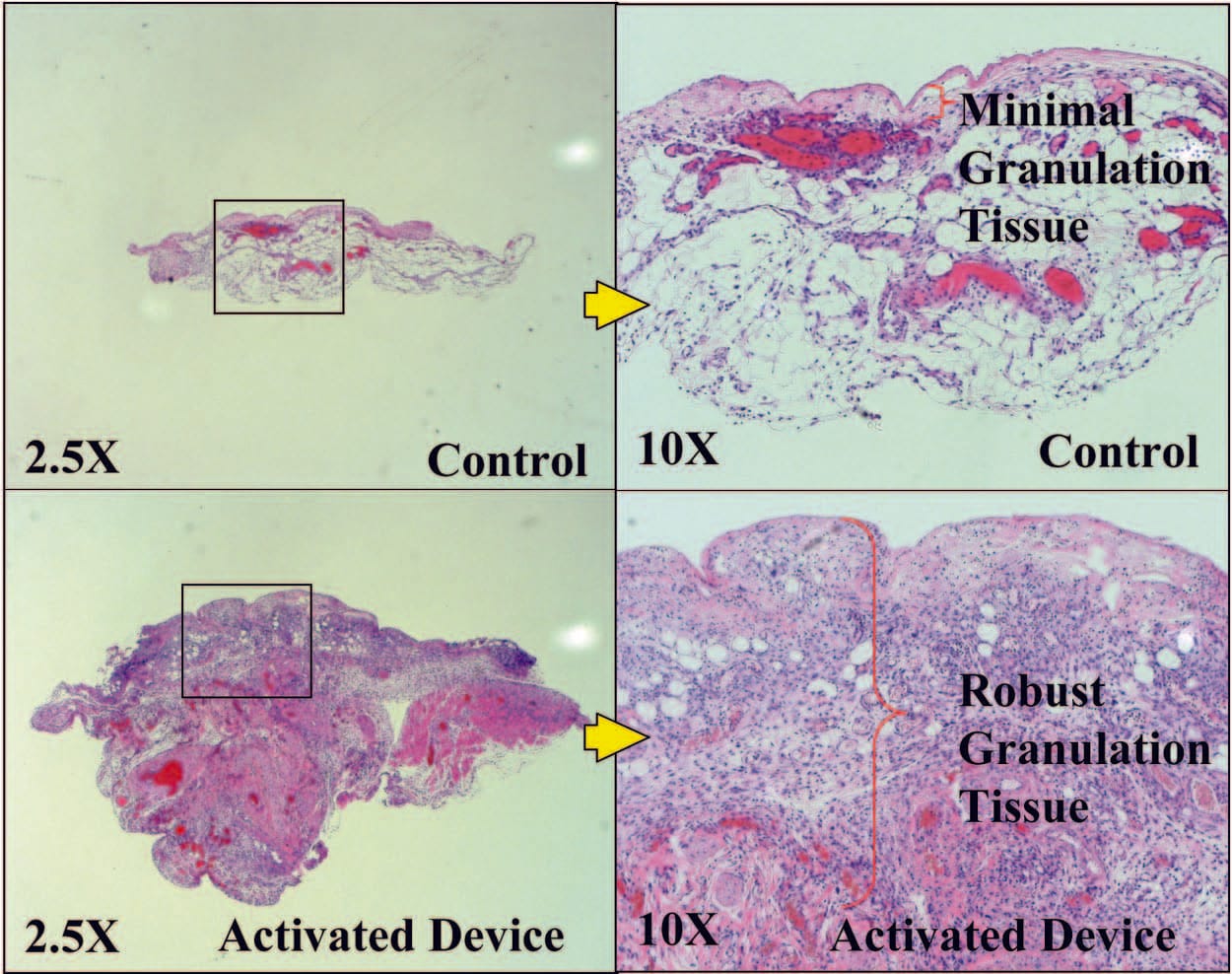
Fig. 8. Representative histologic sections demonstrating increased granulation tissue with SNaP treatment on postoperative day 4.
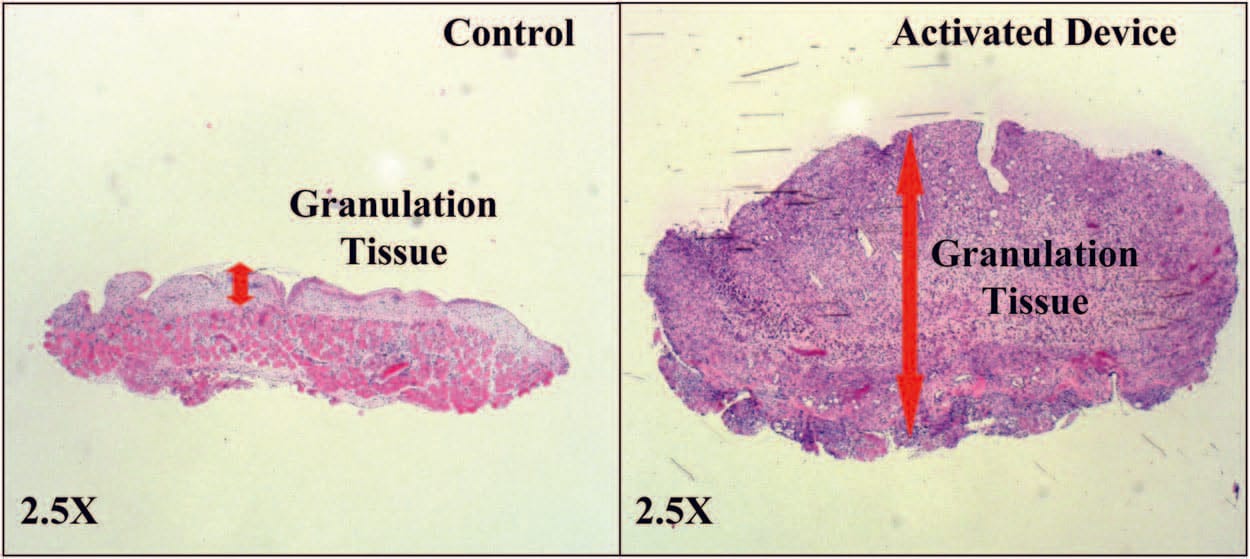
Fig. 9. Representative histologic sections demonstrating increased granulation tissue with SNaP treatment on postoperative day 7.
There were no fatalities, wound infections, or other significant complications from treatment in any of the animal studies. However, the DuoDERM seal did fail in two rat subjects (on postoperative days 3 and 6). The seal was reestablished with the addition of Tegaderm and paper tape to the area of dressing dehiscence.
DISCUSSION
In this study, a novel, ultraportable negativepressure wound therapy system was evaluated. The mechanical testing experiments demonstrated that the SNaP System delivers steady negative pressure to wound beds with and without exudate present in a fashion similar to the vacuum-assisted closure device. The mechanical stress and strain patterns produced by the SNaP System were also comparable to those created by the vacuum-assisted closure device. Thus, from a pressure delivery standpoint, the negative pressure delivered by both systems was essentially identical. Based on these findings alone, the SNaP System would be expected to deliver the same functional benefits to a wound as the vacuum-assisted closure device. To test this hypothesis, the SNaP System was evaluated in vivo using a rat open wound model. The SNaP System delivered effective negative-pressure wound therapy, evidenced by increased granulation tissue formation and faster healing, consistent with published results using vacuum-assisted closure in the same animal model.3
Although human efficacy data are still needed for the SNaP System, and although foam is currently not recommended for use with the SNaP System, the biomechanical and in vivo data suggest that the SNaP System may have efficacy equal to that of vacuum-assisted closure for some wounds. This study was performed in a rodent model, and many differences exist between acute rodent wound healing and wound-healing problems found in humans. In addition, only a single pressure level, –125 mmHg, was tested in vivo. Other pressure levels may have worse, equal, or better wound repair outcomes. However, the potential benefits of a silent, disposable, less expensive, and less cumbersome negative-pressure wound therapy system may prove to be valuable to clinicians.
ACKNOWLEDGMENTS
This work was supported by Spiracur, the Oak Foundation, and the Hagey Laboratory for Pediatric Regenerative Medicine. Portions of the experiment (micro–computed tomographic strain evaluation) were performed at the Stanford Center for Innovation in In Vivo Imaging with collaboration from Dr. Timothy Doyle (Stanford Center for Innovation in In Vivo Imaging). Spiracur and SNaP are trademarks of Spiracur, Inc. All rights reserved.
REFERENCES
- Saxena V, Hwang CW, Huang S, Eichbaum Q, Ingber D, Orgill DP. Vacuum-assisted closure: Microdeformations of wounds and cell proliferation. Plast Reconstr Surg. 2004;114: 1086–1096; discussion 1097–1098.
- Argenta L, Morykwas MJ. Vacuum-assisted closure: A new method for wound control and treatment. Clinical experience. Ann Plast Surg. 1997;38:563–577.
- Isago T, Nozaki M, Kikuchi Y, Honda T, Nakazawa H. Effects of different negative pressures on reduction of wounds in negative pressure dressings. J Dermatol. 2003;30:596–601.
- Armstrong DG, Lavery LA. Negative pressure wound therapy after partial diabetic foot amputation: A multicentre, randomised controlled trial. Lancet 2005;366:1704–1710.
- Vuerstaek JD, Vainas T, Wuite J, Nelemans P, Neumann MH, Veraart JC. State-of-the-art treatment of chronic leg ulcers: A randomized controlled trial comparing vacuum-assisted closure (V.A.C.) with modern wou
nd dressings. J Vasc Surg. 2006;44:1029–1037; discussion 1038. - Morykwas MJ, Simpson J, Punger K, Argenta A, Kremers L, Argenta J. Vacuum assisted closure: State of basic research and physiologic foundation. Plast Reconstr Surg. 2006;117 (7 Suppl):121S–126S.
- Wilkes R, Zhou Y, Cunningham K, Kieswetter K, Haridas B. 3D strain measurement in soft tissue: Demonstration of a novel inverse finite element model algorithm on MicroCT images of a tissue phantom exposed to negative pressure wound therapy. J Mech Behav Biomed Mater. 2009;2:272–287.
- Wilkes R, Zhou Y, Kieswetter K, Haridas B. Effects of dressing type on 3D tissue microdeformations during negative pressure wound therapy: A computational study. J Biomech Eng. 2009;131:031012.
- Orgill DP, Manders EK, Sumpio BE, et al. The mechanisms of action of vacuum assisted closure: More to learn. Surgery 2009;146:40–51.
- Morykwas MJ, Faler BJ, Pearce DJ, Argenta LC. Effects of varying levels of subatmospheric pressure on the rate of granulation tissue formation in experimental wounds in swine. Ann Plast Surg. 2001;47:547–551.


