E.A. Balint*, C.J. Gatt, Jr., M.D., M.G. Dunn, Ph.D. Department of Orthopaedic Surgery, UMDNJ – Robert Wood Johnson Medical School New Brunswick, NJ 08903
ABSTRACT
It is well accepted that the loss of meniscal function due to surgical removal is a significant factor in the onset of degenerative osteoarthritis of the knee. Few viable alternatives exist for the replacement of significantly damaged tissue. In this study, two potential designs for meniscus scaffolds were compared with a focus on the mechanical protection they provide the articular surfaces and their overall biological incorporation into synovial tissue. Three scaffold designs (no fiber reinforcement, 500 fiber scaffold, and 1,000 fiber scaffold) were mechanically loaded in compression at a load of either 100 or 250N. Pressure sensitive film was employed to determine the pressure distribution profile on the tibial plateau after loading. Portions of scaffolds were also implanted into the synovium of the rabbit knee in a non-functional capacity for either 4 or 8 weeks. It was found that the presence of fiber reinforcement in the scaffold increased the contact area on the tibial plateau and decreased the high peak loads for the 100 N loading condition. No differences in biological incorporation were found between the two fiber reinforced scaffolds. Both scaffolds were infiltrated by cells by 4 weeks and tissue deposition was observed. Based on these results, the fiber reinforced meniscus scaffold design is a feasible design for the replacement of significantly damaged meniscal tissue. Further evaluation and optimization are currently underway.
1. INTRODUCTION
The menisci of the knee joint are two C-shaped discs of fibrocartilage found between the long bones of the leg which are critical for proper joint function and health. They provide protection to the articular cartilage by transmitting loads through the joint, distributing high peak stresses evenly on the underlying surfaces, providing shock absorption, and aiding in joint lubrication [1]. The tissue is comprised primarily of collagen (type I) fibers arranged in a circumferential pattern. When the tissue is loaded axially (i.e. weight-bearing on the knee) it is compressed and extruded from the joint capsule. The circumferentially arranged fibers resist this extrusion, thus generating forces in the tangential direction. In essence, an axial compressive load is converted to a circumferential tensile load. Maintenance of healthy joint tissue is dependent on this mechanical relationship.
The importance of the meniscus to overall joint health can best be seen when the tissue is damaged or removed. Investigators have shown the strong correlation between removal of significant amounts of meniscal tissue with degraded joint function as well as the onset of degenerative osteoarthritis of the knee [2-4]. Unfortunately, the menisci are vulnerable to damage during any physical activity which places relatively high translational and/or rotational stresses on the knee (i.e. military training, military activities, sports activities). Additionally, isolated traumatic incidents such as car accidents or falls can cause severe damage to the tissues of the knee. Due to its limited blood supply, the meniscus has poor healing capabilities and injuries to the tissue often require surgical intervention [5, 6].
One type of alternative treatment being explored by investigators is the use of biocompatible, resorbable scaffolds to replace damaged meniscal tissue [7-11]. Several types of scaffolds made up of collagen and various polymers are currently being developed. While some of these devices have shown promising preliminary data, none have been widely accepted by the Orthopaedic community. Many of these scaffolds lack the mechanical strength and integrity to take over the load-bearing role of the meniscus. Furthermore, the amorphous structure of these devices – while conducive to cellular ingrowth – does not provide the appropriate mechanical environment for the formation of new, organized tissue which resembles that of the normal meniscus.
Our current design, based on the microstructure of a normal meniscus, is that of a chemically crosslinked collagen sponge reinforced with synthetic polymer fibers arranged in an organized manner. Collagen is an attractive biomaterial for musculoskeletal tissue engineering due to its unique biochemical and mechanical properties [12]. Its high cellular affinity makes it an ideal coating material for synthetic polymers, which lack cell-recognition signals. Furthermore, the mechanical strength, degradation profile, and porosity of collagen implants can be controlled by various methods of crosslinking. The fiber component of this scaffold is made of a biocompatible and biodegradable, tyrosine derived synthetic polymer called poly(desaminotyrosyl-tyrosine dodecyl dodecanoate)(12,10), or p(DTD DD), or poly(12,10) [13]. Compared to other polymer fibers considered, poly(12,10) fibers have a relatively high ultimate stress and low modulus of elasticity, making this type of fiber an ideal candidate for applications where high stresses and strains are likely to be encountered.
What distinguishes this design from others is its fiber reinforcement. An amorphous structure implanted at the site of meniscal resection is not designed to adequately transmit compressive loads. However, a fiber reinforced structure can experience axial compressive loads and convert them to circumferential tensile loads, thus decreasing the stress on the articular cartilage. To our knowledge, no other studies have been completed on fiber reinforced analogs for the application of a meniscal scaffold. We hypothesize that this structure would allow the cells to experience the same types of mechanical stimuli they would in a normal meniscus, thus promoting neo-tissue growth which resembles that of normal meniscal tissue. A biocompatible implant that can support regeneration of meniscal tissue would restore the load bearing function of the damaged meniscus and prevent the degeneration of the joint and the associated loss of function. If successful, this would have significant impact on the long-term outcome of a total or subtotal meniscectomy.
In this study, two potential fiber reinforced scaffold designs varying in fiber density were explored. The objectives of this study were to first characterize the mechanical properties of the meniscus scaffolds, with an emphasis on the effect of the scaffold on the underlying articular surfaces. Second, a non-functional in vivo evaluation was performed in a rabbit knee to evaluate the biocompatibility of the device implanted in the harsh synovial environment.
2. MATERIALS AND METHODS
Scaffolds were fabricated by wrapping a continuous length of this fiber around a series of pins arranged in a semicir
cular formation on a base plate. A 1% collagen dispersion was then injected into the resultant weave, frozen rapidly in an ethanol/dry ice bath, and then lyophilized. The pins were removed and a length of fiber was repeatedly weaved around the periphery to provide additional structural integrity. The collagen matrix was then crosslinked by submersion in a solution of 1-ethyl-3-(dimethyl aminopropyl) carbodiimide (EDC) as per a protocol previously used in our lab [14]. The result is a chemically crosslinked collagen sponge reinforced with resorbable, synthetic polymer fibers (Figure 1).
Two fiber reinforced scaffold designs were evaluated in this study. One scaffold was fabricated by making 500 passes of the fiber (MS500), the other by making 1,000 passes (MS1000). For the mechanical evaluation, a 100% collagen sponge was also fabricated for a non-fiber reinforced control (MSCOLL).
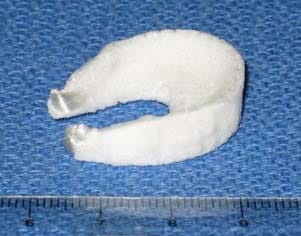
Figure 1: Picture of meniscus scaffold. Polymer fibers are wrapped continuously to provide two strong anchor points for attachment of the scaffold to the tibial plateau.
2.1 Mechanical Evaluation
The purpose of this experiment was to evaluate the ability of the scaffold to distribute loads across the tibial plateau during compressive loading. Four loading conditions were explored in this experiment: (1) no scaffold (n=6), (2) 100% collagen sponge scaffold, MSCOLL (n=6), (3) a 500 fiber scaffold, MS500 (n=6), and (4) a 1,000 fiber scaffold, MS1000 (n=6). Due to the novelty of this scaffold design, a custom-made jig was fabricated for use in this experiment. This jig was constructed using portions of a sheep femur and tibia embedded in polyurethane. Briefly, the upper and lower three inches of a tibia and femur were harvested and cleaned to remove any soft tissue, and then embedded with polyurethane glue. The midline was drilled out and a threaded rod was secured into it with polyurethane glue. This threaded rod fits into aluminum frames which attach to mechanical testing system. Two holes were drilled into the tibial plateau at the insertion sites of the medial meniscus. These holes were used to secure the meniscus scaffold to the tibial plateau during testing. Pressurex® film (Sensor Products Inc., Madison, NJ) was used to characterize the distribution and magnitude of compressive loads on the tibial plateau. This pressure sensitive film releases a red dye when mechanically loaded. The shade of the dye can be correlated to a specific pressure, with dark shades corresponding to higher pressures, and vice versa. Data from the film was quantified using the Topaq® Tactile Force Analysis System (Sensor Products Inc., Madison, NJ).
The tibial and femur attachments were mounted in the Instron at a 45° angle, as this represents a knee angle at which significant stresses are placed on the normal meniscus. All mechanical tests were performed at a slow crosshead speed to minimize any viscoelastic effects from the hydrated scaffold. Scaffolds were loaded onto the tibial attachment of the testing jig and secured through the horn tunnels with suture and a polypropylene button (Figure 2). A section of Pressurex® film was placed between the tibia and the scaffold. A compressive load of 100N was applied to the tibia/scaffold construct and then released. The pressure sensitive film was changed out and then a 250N load was applied. This was done for all fiber reinforced scaffolds. However, the 100% collagen scaffolds ruptured at low loads, so they were either loaded at 100N or 250N only. For a negative control, pressure film was loaded onto the tibia and then loaded at either 100 or 250 N in the presence of no scaffold. From the pressure sensitive film data, the contact area on the tibia and the area undergoing relatively high peak stresses were calculated. All results were statistically analyzed using a two-way ANOVA with pairwise comparisons by the Student-Newman-Keuls method.
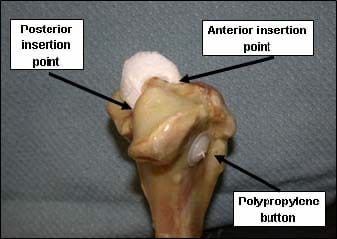
Figure 2: The meniscus scaffold is anchored to the tibial plateau by attaching suture to its horns and feeding it through two bone tunnels. The suture is then secured to the opposite side of the bone with a polypropylene button.
2.2 Non-Functional In Vivo Evaluation
If the implant is to degrade and eventually be replaced by fibrocartilaginous tissue that functions as a meniscus, it must allow for cellular infiltration, proliferation, protein synthesis, and protein deposition. The purpose of this experiment is to evaluate the biologic response to a fiber reinforced meniscus scaffold implanted intraarticularly. Two scaffold designs were studied, MS500 (n=5) and MS1000 (n=5). Two sections were cut out from each scaffold; one for a 4 week time point, the other for an 8 week.
Both knees of each rabbit were used in this study. The MS500 implant was implanted into the left and the MS1000 implant in the right. The surgeon created a mid-line incision over the knee joint, followed by a sharp dissection carried down through the subcutaneous tissue to the extensor mechanism. A medial parapatellar arthrotomy was made, creating a pocket for the implant between the muscle and the medial aspect of the knee joint (Figure 3a). The implant was placed in the pocket and secured with suture (Figure 3b). This surgical procedure allows for the implant to be exposed to the synovial fluid without impeding joint function or being mechanically stressed.
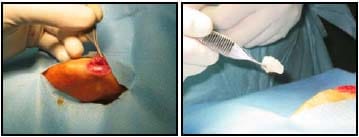
Figure 3a/b: A parapatellar incision was made to create a pocket which allows for exposure to synovial fluid. The implant is placed in the pocket and secured with suture.
After four or eight weeks post-operation, the rabbits were euthanized and the scaffold section along with 2-3 mm of the surrounding tissue was excised and fixed in Carson’s Buffered Formalin. Each section was then processed histologically and stained with either haematoxylin and eosin (H&E) or Masson’s Trichrome. Slides were graded blindly by a university pathologist on two separate dates for inflammation (lymphocyte, eosinophil, and multi-nucleated giant cell), neo-tissue formation, vascular tissue ingrowth, and scaffold degradation. Scores from each day were averaged and analyzed statistically with a two-way ANOVA. All pairwise comparisons were accomplished with the Student-Newman-Keuls method.
4. RESULTS
3.1 Mechanical Evaluation
Figure 4 shows a representative picture of the pressure distribution profile on the tibial plateau and corresponding histogram for each loading condition. Visually, it can be seen that the presence of a fiber reinforced scaffold helped to distribute the compressive load on the articular surface during both simulated loading conditions. For all scaffold and loading conditions, the total contact area was quantified in Figure 5. From this data, it can be seen that the addition of fiber reinforcement resulted in an increase in the total contact area during both loading conditions. Significant differences (*) were found between all groups with the exception of the MSCOLL and ‘No Scaffold’ groups.
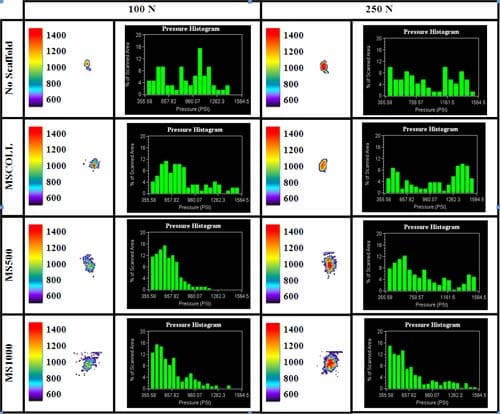
Figure 4: Contact stresses on tibial plateau after 100N or 250N loading for each scaffold condition. The corresponding histogram shows the pressure distribution for each test run.
For the 100N loading condition, a significant decrease in the area experiencing relatively high stress (> 1,200 psi) was found for the fiber-reinforced scaffolds. Furthermore, the fiber reinforced scaffolds had a greater percentag
e of the total contact area under relatively low stress (< 750 psi). For the 250N loading condition, the area experiencing high stresses remained essentially unchanged for all scaffold conditions. However, the corresponding histograms show that the percentage of area which underwent low stress increased significantly with the addition of fiber reinforcement.
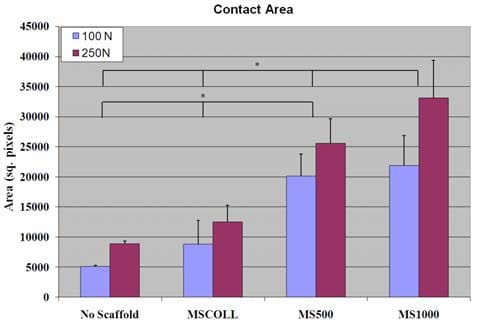
Figure 5: Total contact area for each scaffold and loading condition. Significant differences are noted with a *.
3.2 Non-Functional In Vivo Evaluation
Figure 6 shows a typical picture of each scaffold group at each time period at 100x magnification. Overall observations noted by the pathologist were (1) the majority of lymphocyte inflammation was due to the collagen sponge portion of the scaffold, (2) the presence of eosinophils was correlated with the presence of lymphocytes (3) multi-nucleated giant cells (MNGCs) were associated with the fiber portion of the scaffold, (4) most new collagen growing was found around the MNGCs.
Figure 7 shows comparisons between average scores for both scaffold design groups at 4 and 8 weeks post-op. With regards to lymphocyte inflammation, the presence of eosinophils, and the vascularity of the neo-tissue, there were significant differences between the 4 and 8 week groups as a whole, as well as differences within each scaffold type between the 4 and 8 week groups. With regards to scaffold degradation, there was a significant difference between the 4 and 8 week groups as a whole, but only a difference between the 4 and 8 week groups within the MS1000 scaffold group. No other differences were noted.
4. DISCUSSION
Healthy, functional meniscal tissue is essential for the maintenance of the articular cartilage of the knee. By distributing stresses evenly on the tibial and femoral surfaces, high peak loads detrimental to cartilage tissue are avoided. Since only about a third of the meniscus is vascularized, the tissue has a limited healing potential. As such, damaged tissue often requires surgical removal, which leaves the patient at a much higher risk for degenerative changes in the knee. Currently, there are no widely accepted treatment alternatives to significant meniscal removal. The use of meniscal allograft has had limited success, and its long-term benefits have yet to be proven. The use of resorbable scaffolds is currently being explored by several investigators [7-11]. While some of these devices have shown promising preliminary data, none have been widely accepted by the Orthopaedic community [15]. Many of these scaffolds lack the mechanical strength and integrity to take over the load-bearing role of the meniscus. Furthermore, the amorphous structure of the devices – while conducive to cellular and tissue ingrowth – does not provide the appropriate framework for the formation of new, organized tissue which resembles that of the normal meniscus.
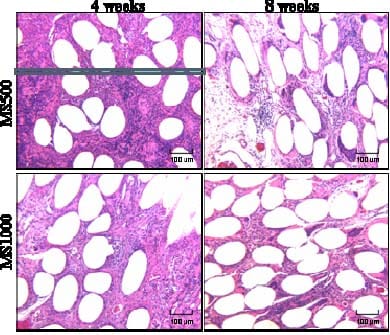
Figure 6: Representative pictures of MS500 and MS1000 scaffold designs at 4 and 8 weeks post-op, stained with H&E, at 100x magnification.
Our lab is currently developing a meniscal scaffold which addresses the complex mechanical behavior of the meniscus. Two types of fiber reinforced scaffolds varying in the amount of the polymer fiber used were explored in this study. In the first aim of this study, the ability of the fiber reinforced scaffolds to distribute loads on the tibial plateau was evaluated and compared to similar loading conditions in the presence of no scaffold or a non-fiber reinforced collagen scaffold. The 100N and 250N loads were chosen to simulate light-to-moderate loading of a quadruped’s meniscus. The benefits of fiber reinforcement were clearly seen by the significant increase in contact area and decrease in high peak loads as compared to the no scaffold loading (comparable to a knee which has undergone a total meniscectomy) and the MSCOLL loading (comparable to the non-fiber-reinforced scaffold being explored by other investigators). This suggests that the scaffold was able to mimic the biomechanical behavior of the normal meniscus. When the scaffold was axially loaded in compression, it was extruded from the joint. The anterior and posterior anchor points resisted this extrusion, forming circumferentially oriented hoop stresses, thus dissipating the high stresses and distributing the load more evenly on the tibial plateau. Further mechanical evaluation is currently underway to quantify how much of the axial compressive load is converted to a circumferential tensile load.
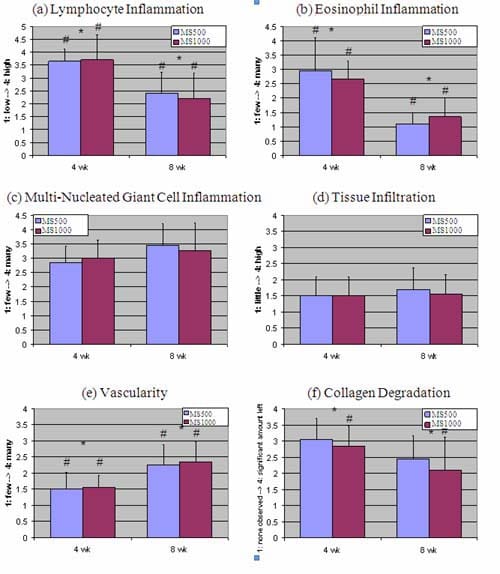
Figure 7: Average scores for scaffolds. (a) Level of lymphocyte inflammation, (b) presence of eosinophils, (c) presence of multi-nucleated giant cells, (d) level of tissue infiltration, (e) vascularity, (f) degradation of the implanted collagen matrix. A * denotes a significant difference between time groups. A # denotes a significant difference between time groups within a scaffold group.
A non-functional in vivo evaluation was completed due to its relative simplicity and the need for a small as opposed to a large animal model. This experiment allowed for the evaluation of the biological response to the scaffold while exposed to the harsh synovial fluid. Histological analysis by a pathologist revealed three main types of inflammatory cells: lymphocytes (Figure 7a), eosinophils (Figure 7b), and multi-nucleated giant cells (Figure 7c). Lymphocyte and eosinophil inflammation was associated most with the collagen matrix portion of the scaffold. The lack of difference between lymphocyte or eosinophil inflammation between scaffold types suggests that only a small amount of the matrix is needed to initiate the response seen. It is also possible that any differences between scaffolds could have been seen at an earlier time point, but by four weeks, the lymphocyte and eosinophil inflammation had leveled off to approximately the same. This also may hold true for the presence of multi-nucleated giant cells (MNGCs), which were primarily observed around the fibers. By four weeks, MNGCs had formed around fibers present in the scaffolds. The reason no difference was detected between scaffold types (despite the obvious difference in fiber count) was that the presence of MNGCs was graded as an average per field of view compared to the surrounding structures, and not as a total number of MNGCs per scaffold section. Figure 6 shows that the fields of view for both MS500 and MS1000 scaffolds were quite similar, so detecting differences in the total number of MNGCs is unrealistic.
The level of tissue infiltration into the scaffolds was not significantly different over time or between scaffold types (Figure 7d). The level of inflammation observed suggests both scaffold types were in the granulation stage of the wound healing process. The new tissue formed primarily around the MNGCs and was most likely the result of an encapsulation response to the slowly degrading fibers. In a non-functional environment, this neo-tissue would have been irregularly arranged and lacking of substantial mechanical strength. It is possible that the low level of tissue infiltration may have been due to the lack of mechanical stimulation of the scaffold. In a functional model, where the fibers are experiencing periodic loading and unloading, the tissue formed around the fibers may mature earlier. The persistent inflammation of the scaffold may also contribute to the lack of high tissue infiltration. This inflammation (Figures 7a-b) is directly related to the degradation of the implanted collagen matrix (Figure 7f). As such, the presence of high amounts
of collagen scaffold may delay the deposition of the neo-tissue. However, the collagen portion of the scaffold is considered necessary due to its chemotactic properties. In the environment of the knee, a substance which attracts cells involved with the wound healing process is necessary. So while the collagen matrix may result in an early inflammatory response, its presence is necessary to get the cells needed to infiltrate into the scaffold and begin the process of incorporation. The vascularity of the scaffolds increased over time, but did not differ between scaffold types. For the scaffolds to begin the integration process into the body, the infiltration of vascular tissue is necessary. Both scaffold types supported the infiltration of blood vessels into the scaffold, and by 8 weeks, these vessels were more prominent than at 4 weeks. The presence of less collagen matrix and more tightly packed fibers in the MS1000 scaffold did not impede the growth of vascular tissue as compared to the MS500 scaffolds.
Collectively, these results show feasibility of this new approach to reconstruct meniscal tissue after severe meniscus injury. The in vivo experiment demonstrates that cells rapidly infiltrate the scaffold and begin laying down tissue as early as 4 weeks post-implantation. Furthermore, from a biomechanical standpoint, the reinforced scaffold behaves similarly to the normal meniscus under loading. This is especially important during the tissue remodeling process, where cells synthesize and lay down proteins based on the type of mechanical load they experience. In this case, a cell attached to the circumferential oriented fiber may experience tensile loads, and begin laying down type I collagen in a circumferentially organized manner – similar to that of the normal meniscus. Current studies are focused on further optimization of mechanical and biological properties of these novel meniscus replacement scaffolds, as well as functional in vivo evaluations in a large animal model.
CONCLUSION
Injuries represent the largest health problem faced by the military today. The costs associated with these injuries may include loss of manpower, rehabilitation costs, waste of training time/money, cost to retrain members as replacements, hospitalization costs, disability costs, etc [16-19]. Injuries to the knee are well documented in the military population. These injuries can occur on-duty, especially for members in a career field where strenuous activities are the norm (i.e. infantry, special operations, security/military police, etc.), or during off-duty activities, such as regular physical training. Most knee injuries involve the menisci in some fashion and depending on the severity of the injury, complete derangement of meniscal tissue in not uncommon. A damaged meniscus can severely impair the normal activity of a military member. The severe pain associated with damaged meniscal tissue as well as osteoarthritis in the knee can have a debilitating effect on the ability of servicemen/servicewomen to perform their jobs. Thus the operational capability of a military member with an injured meniscus can be inhibited in the short and long term. A viable tissue engineered meniscus replacement would offer a solution to service members who have moderate to severe meniscal damage, allowing them to return to duty even after a significant knee injury. This study has demonstrated the feasibility of a fiber-reinforced meniscus scaffold for the replacement of significantly damaged meniscal tissue. The scaffold possesses the structure to 1) respond to the complex biomechanical loading environment of the knee, and 2) promote cellular and tissue infiltration into the scaffold.
ACKNOWLEDGEMENTS
The p(DTD DD) polymer and fiber were provided by the New Jersey Center for Biomaterials (Joachim Kohn, Ph.D. and Sanjeeva Murthy, Ph.D.). This work was supported by grants to CJG from the National Institutes of Health R21 AR052118 and the Center for Military Biomaterials Research (CeMBR) grant # W81XWH042003. The review of the histology slides was completed by Parisa Javidian, M.D., Associate Professor, Pathology and Laboratory Medicine, UMDNJ.
REFERENCES
- Arnoczky, S.P. and C.A. McDevitt, The Meniscus: Structure, Function, Repair, and Replacement, in Orthopaedic Basic Science: Biology and Biomechanics of the Musculoskeletal System, J.A. Buckwalter, T.A. Einhorn, and S.R. Simon, Editors. 2000, American Academy of Orthopaedic Surgeons. p. 531-545.
- Casscells, S.W., The torn or degenerated meniscus and its relationship to degeneration of the weight-bearing areas of the femur and tibia. Clin Orthop Relat Res, 1978(132): p. 196-200.
- Fairbank, T., Knee joint changes after meniscectomy. J Bone Joint Surg (Br), 1948. 30: p. 664-70.
- Krause, W., et al., Mechanical changes in the knee after meniscectomy. J Bone Joint Surg, 1976. 58(A): p. 599-604.
- King, D., The healing of semilunar cartilages. Clin Orthop Relat Res, 1936(252): p. 4-7.
- Arnoczky, S.P., Gross and Vascular Anatomy of the Meniscus and Its Role in Meniscal Healing, Regeneration, and Remodeling, in Knee Meniscus: Basic and Clinical Foundations, V.C. Mow, S.P. Arnoczky, and D.W. Jackson, Editors. 1992, Raven Press: New York. p. 1-14.
- Buma, P., et al., Tissue engineering of the meniscus. Biomaterials, 2004. 25(9): p. 1523-32.
- Heijkants, R.G., et al., Design, synthesis and properties of a degradable polyurethane scaffold for meniscus regeneration. J Mater Sci Mater Med, 2004. 15(4): p. 423-7.
- Steadman, J.R. and W.G. Rodkey, Tissue-engineered collagen meniscus implants: 5- to 6-year feasibility study results. Arthroscopy, 2005. 21(5): p. 515-25.
- Tienen, T.G., et al., Replacement of the Knee Meniscus by a Porous Polymer Implant: A Study in Dogs. Am J Sports Med, 2005.
- Stone, K.R., et al., Regeneration of meniscal cartilage with use of a collagen scaffold. Analysis of preliminary data. J Bone Joint Surg Am, 1997. 79(12): p. 1770-7.
- Stenzel, K.H., T. Miyata, and A.L. Rubin, Collagen as a biomaterial. Annu Rev Biophys Bioeng, 1974. 3(0): p. 231-53.
- Bourke, S.L. and J. Kohn, Polymers derived from the amino acid L-tyrosine: polycarbonates, polyarylates and copolymers with poly(ethylene glycol). Adv Drug Deliv Rev, 2003. 55(4): p. 447-66.
- Caruso, A.B., A Collagen Fiber Tissue Engineering Scaffold for Anterior Cruciate Ligament Reconstruction, in Dept. of Biomedical Engineering/Dept. of Orthopaedic Surgery. 2004, Rutgers University/University of Medicine and Dentistry of New Jersey: New Brunswick. p. 148.
- Buma, P., T. van Tienen, and R. Veth, The collagen meniscus implant. Expert Rev Med Devices, 2007. 4(4): p. 507-16.
- Atlas of injuries in the United States Armed Forces. Mil Med, 1999. 164(8 Suppl): p. 633 pages.
- Lauder, T.D., et al., Sports and physical training injury hospitalizations in the army. Am J Prev Med, 2000. 18(3 Suppl): p. 118-28.
- Kaufman, K.R., S. Brodine, and R. Shaffer, Military training-related injuries: surveillance, research, and prevention. Am J Prev Med, 2000. 18(3 Suppl): p. 54-63.
- Cox, K.A., et al., Prior knee injury and risk of future hospitalization and discharge from military service. Am J Prev Med, 2000. 18(3 Suppl): p. 112-7.