Group 18: Thomas Kopin (EE)
Kevin Lee (EE)
Additional Members: Jason Bulaclac (ME)
Naiem Sulaimanee (ME)
Daniel Taskalos (Physics)
Advisors: Dr. Yong Shi
Dr. Stuart Tewksbury
Abstract
With stroke being the third leading cause of death in the United States, there is a need for the discovery of alternate treatments that can both save and improve the lives of those afflicted by a stroke. The purpose of the design team is to design, build, and test a macro model device that can mechanically ease the effects of a stroke by removing or breaking down the blood clot that causes a stroke. To solve the problem the team researched similar devices and analyzed their sources of failure. After researching, the team was able to brainstorm five designs and narrowed them down to one design that we agreed to solely pursue to the next stages of development. The design that will be used is a vacuum strainer device that will pierce the blood clot, vacuum the clot into the device, and break it up into smaller pieces where it can be reintroduced into the blood stream. If the team is able to prove our concept, it can potentially be manufactured and used by doctors to help stroke victims all over the world.
Introduction
Stroke is the third leading cause of death in the United States and will occur in 700,000 Americans this year, of these 700,000, over 163,000 of them will die. Ischemic stroke is when a blood clot, called a thrombus, blocks blood in a cerebral artery from entering the brain causing brain cells to die. Ischemic stroke is the most common type of stroke and accounts for 85% of all strokes. Time is very important when treating a stroke since every second that passes, 32,000 brain cells die. Treatments of ischemic stroke include pharmacologic thrombolysis (intravenous drugs that dissolve blood clots) and mechanical thrombectomy (removing the clot mechanically).
The objective of this project is to design a MEMS (microelectromechanical system) device to mechanically remove or break apart a blood clot in the cerebral artery in stroke patients. The device will need to completely relieve the effects of the stroke and replenish blood to the brain in a small amount of time. A macro scale prototype of the device will be built and demonstrated in an in vitro model.
Considering the medical arena we’ve entered with the commencement of this project, the impact of it directly depends on the efficiency of our process. The removal of blood clots from a patient’s vessels could be a life or death situation. With the current method of injecting tissue plasminogen activator (tPA) to dissolve blood clots, the probability of recurrence makes it a short term solution with negative long term effects. Our device aims to provide a more complete treatment and reduce the recurrence rate while limiting vessel damage for our patients. The fact that our device can be used in a proactive process (rather than waiting for a removing agent to do the work) makes it all the more significant.
Design Requirements:
- Minimalization of further damage
- Effect on blood flow
- Vessel wall safety
- Ease of use
- Cost of manufacture
- Removal of thrombus
- Destruction of thrombus
- Durability
Several factors need to be considered in the selection of the final design. The criteria are the durability, removal of the blood clot, destruction of the blood clot, cost of manufacture, ease of use, vessel wall safety, effect on blood flow, and minimalization of further damage. The ease of use is concerned with how easy the device is to use and if any additional training is needed to operate. Each concept is broken up into either a design to remove the clot or destroy to clot. Any concept needs to limit the chance of further damage to the body, such as tissue decay or vessel destruction. Cost is of vital importance to ensure that as many patients as possible could benefit from the design’s advantages. We also looked into the existing products to determine their successes and design failures in order to design the best product possible. Below are examples of existing products;
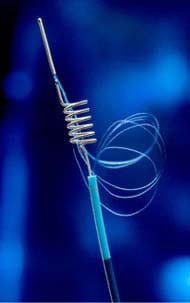
1. Merci Retrieval System
The MERCI (Mechanical Embolus Removal in Cerebral Ischemia) retrieval system is the only device approved by the FDA (Food and Drug Administration) as a treatment for ischemic stroke. The device is introduced into the body using a catheter and navigated though blood vessels to the brain. The end of the device is fitted with a corkscrew where it can be driven though the clot and mechanically remove it. A recent study with the device showed success in 48% of patients where just the device alone was used and the success rate increased to 60% when the device was used in combination with drug therapy.
2. AngioJet System
The AngioJet system is a device that uses saline jets directed back into the catheter to create a low pressure zone at the end of the device to essentially vacuum the clot to remove it from the body. However, the system was designed to remove clots from other arteries other than from the brain.
3. Latis Laser Device
The Latis laser device uses a laser at its tip to heat the clot to the point where it would breakdown. However, clinical trials showed that surgeons were unable to successfully get the device to the brain and further trials were abandoned.

4. Endovascular Photo Acoustic Recanalization Laser
The endovascular photo acoustic recanalization (EPAR) laser uses directs laser energy to the clot where it is absorbed and converted to acoustic energy. The clot is then broken up into small globules by the tip of the device. Clinical trials showed success in 44% of patients were the device was used in conjunction with drug therapy, but only a 15% success rate were the device was used alone. Currently, lack of funding has stopped any further clinical trials.

5. EKOS Ultrasound Device
The EKOS ultrasound device uses a small ultrasound transducer at the tip of the device in combination with drug therapy to dissolve the clot. The ultrasound waves increase the permeability of the clot in order to speed up the effects of the drugs. Clinical trials of this device are currently ongoing, preliminary results show that complete breakdown of the clot takes an average of 46 minutes.
After examining the existing products we determined the advantages and disadvantages of each device:

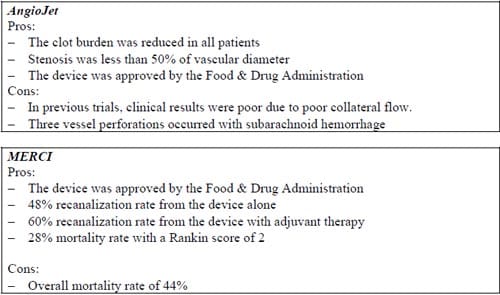
Design Approaches:
With previous research on existing designs and their pros/cons, certain criteria was necessary in developing conceptual designs. One significant consideration was the nature of a blood clot. A blood clot, composed of platelets that exist within blood, has a viscous consistency. Although it blocks the passage way of blood, it does not have a solid form that is easily grasped. Putting this into consideration, various designs were constructed. All the devices would be accompanied by a guide wire which would be directed by a surgeon through a patient via external controls. The location of the device would be monitored via CAT scan.
One design was an “umbrella” like device designed to capture the clot and remove it from the body. The device would initially be collapsed and enclosed by a tip. The surgeon would guide the tip past the clot. Once the device was properly placed, the “umbrella” would inflate, creating a scoop which could then be redirected out the body.
Another design designated for removal is the “jaw” device. Its design is compromised of a clamp like mechanism which encloses the clot within itself. The device would prevent the mass from escape. The surgeon could then pull out the clot via the path entered with the aid of the guide wire.
Another form of treatment is to break up the blood clot into particles minute enough to reenter the bloodstream and no longer prove harmful. A viable option is to use a vacuum strainer; the concept behind this would be to strain the blood clot through a micro-mesh or push the clot through a small opening. To do this, there would need to be some type of encapsulated tip that would be able to push and/or pull the clot through the strain/hole using some type of vacuum.
Applying the same principle of breakdown, a method that would use an active-tip is also a plausible option. This method would involve using minimal vibration induced from an active-tip. At proper frequencies, the tip would be able to induce breakdown of the clot.
A similar method would be using a bristle device. This device would encompass closely to the walls of the blood vessel rotating within the clot and thereby breaking it down. This concept is closely related to a street cleaning brush.
These thus far are the options which have been entertained. A concept comparison matrix is used to determine the overall effectiveness of these ideas based on our criteria. This is of course does not mean that there will we no further emphasis placed on new ideas and possibilities. New concepts are constantly being entertained and these will be evaluated thusly.
Budget
According to our multiple design approaches, we determined that our designs are compromised of two things: the wire, and the tip. The wire will act as the guide to bring the tip to the clot site. The design of the wire will remain the same for each design approach. However, the tip that will either extract or remove the clot will obviously need to change depending upon the approach. Therefore, with this logic in mind, the budget for our different designs will remain very similar.
A final production design has yet to be determined. The group is planning on making a macro model in order to demonstrate the basic premise of the device. The real device will use nitinol wire, which is a common wire used in the production of endoscopes and other medical equipment. The macro model will be made of a wire, which on the macro level should accurately portray the movements of the nitinol at a cheaper price. The simulator will be easy to set up repeatedly for more then one demonstration.
| Macro Model | |
| Computers | $1000 |
| Pressure Sensors | $180 |
| Transparent Tubing | $20 |
| Clot Substance | $5 |
| Simulation Software | $200 |
| Total | $1405 |
Gantt Chart
On our gantt chart we display all of the deliverables and objectives that we wish to meet specifically for the first semester but also for the second semester of Senior Design. The basis of our project is to create an in-vitro model that will display how our project will perform its task. Because this model will remain exactly no matter which conceptual design idea is chosen, we feel that only one gantt chart is necessary to get our point across. All of our conceptual ideas will be designed to perform the same task but possible in a different way. This allows us to keep a general gantt chart that will apply to all designs. As our gantt chart shows we are currently on schedule as a group. We have completed all of the deliverables up till now and are on track to meet all objectives in the future. We have been in close contact with our mechanical advisor in attempt to get a conceptual idea approved for further research.
Conclusions
With stroke being the third leading cause of death in the United States, there is a need for a device that can reduce the overall damage a stroke does to the body. The goal of the group is to design, build, and test a macro scale device that can enter the blood vessels of the body, be navigated to the site of a blood clot in the brain, and either remove or breakdown the clot. Phase I of the project was composed of research of the problem, brainstorming of concepts, and the evaluation of the concepts. The research showed that there have been many attempts to make a device that can rid a blood clot from the brain but only one has been FDA approved. After the research, the group was able to brainstorm five promising ideas of a new device. As we enter phase II of the project, the group has agreed to pursue a vacuum strainer type device to solve the problem of quickly restoring blood flow to the brains of stroke victims. The next steps for the group are to finalize the design and make calculations on how the device will perform.
Since this project is interdepartmental most of the electrical engineering aspects that we will be responsible for have for the most part not been addressed yet. The majority of the project so far has been to come up with a mechanical method to remove the blood clot from the patients veins. Most of the electrical engineering work will be done in designing the model and simulation for the product. We are going to need to design a macro blood vessel which is as close to lifelike as possible. We will need to use sensors and instruments to create an environment similar to the human body. We will also need to use computer software to receive said data and interpret it. We will also be responsible for researching what would have to change with our product in order to shrink it down to a usable device in the field. This research will be done because we do not have the technology or knowledge to create an actual to scale usable model but we do wish to know how it could be done. The electrical engineering role will become very important, especially in the second semester of senior design although we have still played an integral part in getting the group to the point it is at currently.
References
- Khan, Salwa, and Joseph D. Dickerman. “Hereditary Thrombophilia.” Thrombosis Journal 4 (2006). 23 Sept. 2007 .
- Lutsep, Helmi L. ” Mechanical Thrombolysis in Acute Stroke.” eMedicine 2 Nov. 2006. 10 Sept. 2007 < http ://www.emedicine.com/neuro/topic702.htm>.
- “Stroke Facts.” St. John’s Hospital. 25 Sept. 2007. < https://www.stjohns. org/services/stroke_center/stroke_facts.aspx>.
- Marieb, Elaine N. and Katja Hoehn. “Cerebrovascular Accidents.” Human Anatomy and Physiology. 7th ed. Person Education, Inc. 2007. p. 468.


